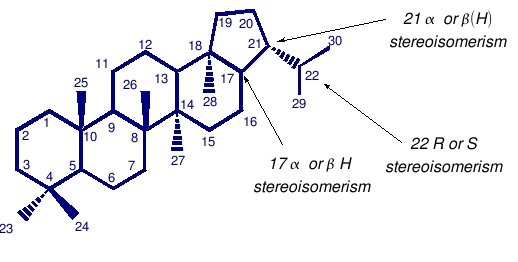
It is often said that hopanoids are the ‘most abundant natural products on Earth.’ Most commonly, hopanoids are found in select groups of Bacteria, all of which are aerobic. Although they are known to be synthesized by a wide variety of cultured aerobic bacteria, there does not to be any obligate requirement for oxygen in their biosynthesis and cyclization of squalene to a pentacyclic triterpanoid with a hopane skeleton. Therefore, hopanoid synthesis might also be possible in anaerobes. The characteristic base structure of a hopane (the degraded and saturated version of a hopanoid), shown above, has four cyclohexane rings and one cyclopentane ring, and might have a side chain emerging from C30. Prokaryotic cells utilize certain types of hopanoids in their cell walls (as opposed to eukaryotes, who use certain types of sterols).
Stereioisomerism
During diagenesis and catagenesis, the biological stereospecificity of hopanoids, particularly at C-17 and C-21 is usually lost, and isomers are generated (see above for numbering). The term alpha beta hopane is commonly used as short-hand to denote hopanes with the 17 alpha(H), 21 beta(H) configuration, while alpha alpha hopane would denote 17 alpha(H), 21 alpha(H) stereochemistry. The notation 17 alpha(H) indicates that the hydrogen is located below the plane of the paper, whereas in 17 beta(H) it is above the plane. The prefix ‘nor’, as for example in 30-norhopane, indicates that the molecule is formally derived for the parent structure by loss of the indicated carbon atom, i.e. C-30 is removed from the hopane skeleton. Similarly, bisnor-, dinor- or trinor- hopane indicate that the molecule is derived from the parent structure by loss of two or three carbon atoms at the specified positions. For example, 25, 28, 30 trinorhopane indicates that carbons at those positions were lost from the parent molecule.
Some examples
2-methyl hopane
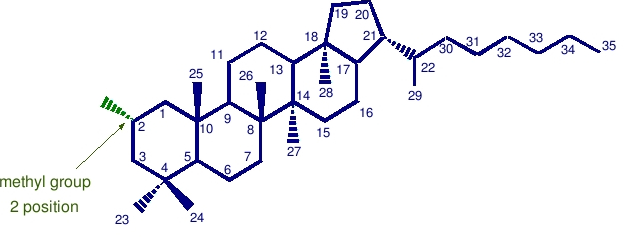
The presence of alkyl substituents on the hopanoid skeleton, like 2-methyl hopane (above) and 3-methyl hopane (below) appears to be limited to specific physiological groups. 2 beta -methylhopanoids are produced by many cyanobacteria and have few other demonstrated sources. Thus, the corresponding sedimentary 2 alpha -methylhopane hydrocarbons are hypothesized to be biomarkers for cyanobacteria. These biomarkers may help to establish the time of the beginning of oxygenic photosynthesis on the primitive earth.
Summons R.E., Jahnke L.L., Logan G.A. & Hope J.M. (1999) 2-Methylhopanoids as biomarkers for cyanobacterial oxygenic photosynthesis. Nature 398:554-57.
3-methyl hopane
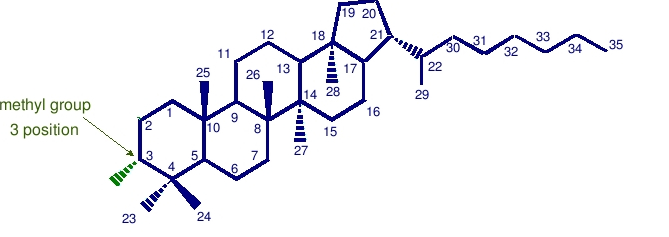
Methanotrophic and acetic acid bacteria biosynthesize a range of 3 beta-hopanoids. The corresponding 3 beta-methylhopane hydrocarbons could be derived from either group of bacteria, but a large 13C depletion observed in their sedimentary sources points to methanotrophic sources being more important. Methanotrophic bacteria may have been among the most primitive forms of life in the early earth.
Bisnorhopane and trisnorhopanes
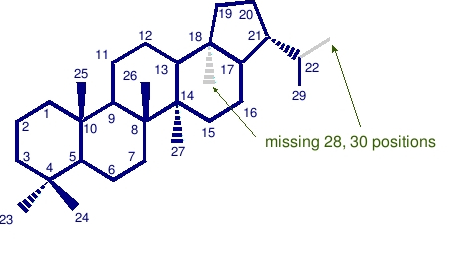
Bisnorhopanes are missing 2 carbon atoms in the positions denoted. Shown here is a 28,30 bisnorhopane, also known as BNH.
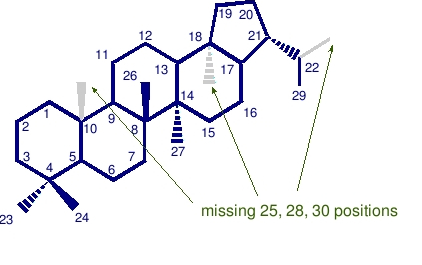
Trisnorhopanes are missing 3 carbon atoms in the positions denoted. Shown here is a 25,28,30 trisnorhopane, also known as TNH.
High concentrations of BNH and TNH are typical of anoxic or euxinic depositional environments. There is strong correlation between the presence of BNH and a sulfidic water column. Their occurence indicates that some bacteria specific to these environments are the ultimate source, but are currently unidentified.
Ts and Tm are specific types of trisnorhopanes (specifically 18 alpha (H) 22,29,30 trisnorhopane and 17 alpha (H) 22,29,30 , respectively). Ts is more stable than Tm, and degrades less during diagenesis and catagenesis. Thus the ratio of Ts to Tm is an indicator of the maturity of a rock.


